Chapter 9: Anger
Anger and the Brain
Amygdala
Like with fear, the amygdala is often activated for anger experiences. Lesions of the amygdala cause changes in social dominance hierarchies, such that previously high-status monkeys become lower in status after the lesion (Rosvold et al., 1954). But, other times, Klüver -Bucy Syndrome (Klüver & Bucy, 1939) in monkeys makes the monkey act tame, even though the situation would typically elicit aggression. Thus, lesions impair the normal anger behavior changes, sometimes resulting in patterns that are opposite the expected patterns for anger. Some recent work is suggesting that the amygdala plays an important role in regulating anger and aggression. Lesions to the human amygdala both increased and decreased aggression (for a review, Haller, 2018; van Elst et al., 2000). Taken together, damage to the amygdala suggests that this structure regulates behavior changes during an anger experience – either by increasing aggressive approach or down-regulating aggression. Some work (Matthies et al., 2012) has found that amygdala volume and trait aggression are negatively correlated. This means that as the amygdala volume decreases, people exhibit greater levels of an aggressive personality trait. Research on mice, voles, hamsters, and even lizards suggests that different parts of the amygdala may be specific to different types of aggression (for a review, see Haller, 2018). For instance, the medial amygdala is activated for male rivalry aggression to acquire dominance (activated with the intruder test discussed in fear), the central amygdala for predatory aggression, and maternal aggression activated both the medial and central amygdala.
One study (Coccaro et al., 2007) compared control participants to participants diagnosed with intermittent explosive disorder (IED), a disorder defined by overly aggressive behaviors. While an fMRI scanned their brain, participants viewed seven of Ekman’s FACS emotional expressions and clicked a button to indicate the gender of the individual in the photo. Results showed that when participants viewed only the ANGRY faces, IED participants exhibited greater activity in the amygdala, but less activity in the orbitofrontal cortex (OFC), compared to control participants. Does this show anger results in an overactive amygdala and underactive OFC? Maybe! But consider which emotions participants may have been experiencing when viewing the ANGRY face.
Orbitofrontal Cortex
The orbitofrontal cortex (OFC) is located in the prefrontal cortex. The OFC is activated when viewing anger facial expressions, highlighting the role of anger perception (Blair et al., 1999), and this activation increases as the intensity of the anger expression increases. Damage to the OFC is associated with 1) impairment in identifying negative, but not positive emotion facial expressions, 2) difficulty identifying the intensity of emotions on facial expressions, and 3) greater reactive aggression (Blair, 2012; Blair & Cipolotti, 2000; Dal Monte et al., 2013; Heberlein, et al., 2008; Marinkovic et al., 2000; Zald & Andreotti, 2010). A recent study (Willis et al., 2014) found that OFC damaged-patients experienced difficulty recognizing emotions of faces in the first 500 ms that a photo was shown but were able to eventually correctly identify the emotion. These findings suggest that impairments may occur at a more nonconscious level. Studies on normal populations have shown that as the OFC shows greater activation to angry faces, people exhibit reduced aggressive behavior (Beyer et al, 2015). These findings would suggest that for normal functioning individuals, when we experience anger, the OFC helps us to regulate our aggressive approach behaviors.
Activation of the OFC is negatively correlated with aggressive behavior. But remember when we view an anger expression on another person’s face, that might cause us to experience fear. Taken together, these findings might suggest that the OFC helps us to perceive anger in others, to down-regulate our anger/aggression, and maybe even to engage in fear avoidance. Paralleling findings on the ventral striatum, lesions to the OFC hinder recognition of anger facial expressions and can also increase aggressive behavior. Said another way, if the OFC is not normally operating, then we might fail to perceive anger and our OFC cannot regulate our anger expression.
In the study discussed above (Coccaro et al., 2007), IED participants reported reduced OFC activation when viewing angry and happy faces, suggesting underactive OFC relates to positive emotions too.
Periaqueductal Gray (PAG) regions of mid-brain
In the 1960’s, Reynolds (1969) accidentally learned that stimulating the PAG in rats allowed him to perform surgery on the rats without using anesthetics. This was the first study to notice the role of the PAG in reducing pain. The PAG also regulates other physiological responses during an emotion, such as the heart rate or blood pressure. Siegel et al. (1999) discusses that electrically stimulating the hypothalamus and PAG results in aggression in cats.
Reward Circuit
The ventral striatum is located in the basal ganglia and is considered part of the brain’s reward circuit. This reward circuit contains high levels of dopamine, so often these structures are activated when people anticipate upcoming rewards. When people recalled and described personal stories of anger, the ventral striatum was activated (Hornak et al., 2003). While describing these stories, participants reported angry subjective feelings, suggesting the ventral striatum is activated when we experience anger. In one study (Hornak et al., 2003), participants recalled times that they felt angry and rated their subjective feelings, all while an fMRI scanned their brain. Findings showed that self-reported feelings of anger were positively correlated with activity in the reward circuit. BUT the problem is that self-reported positive emotions are also positively correlated with activity in the same areas. So, we cannot say that the ventral striatum controls only specific positive or negative emotions. It is more likely that activity is this area is associated with approach emotions. Damage to the ventral striatum (Calder et al., 2004) or interfering with the dopamine receptors (Lawrence et al., 2002) hinders people’s ability to recognize anger in facial expressions. Taken together, these findings indicate the ventral striatum regulates approach behavior and controls ability to perceive anger in others. Furthers, some believe that the ventral striatum may be activated for instrumental aggression, when we are approaching a goal, and may be less related to reactive aggression due to a threat.
Warrior Gene
The warrior gene is the everyday term used to describe the presence of a polymorphism on the enzyme monoamine oxidase A (MAOA). This polymorphism is called low activity (L) variant of monoamine oxidase A (MAOA). A similar genetic mutation was originally discovered in a Dutch family in which many male relatives had a history of violence, extended periods of anger, and low levels of IQ (Bruner, 1993a; for more information, Brunner et al., 1993b). MAOA is an enzyme that controls the levels of other neurotransmitters and hormones. If a person does not have enough MAOA, they could end up with too much of the neurotransmitter or hormone. With the presence of the L variant, people do not have enough MAOA and end up with too much dopamine or testosterone, thus resulting in a tendency for “warrior-like behavior” or aggression. Conversely, with the high (H) variant of MAOA people exhibit adequate or normal levels of MAOA. So, technically, this warrior gene is one cause of an angry or aggressive personality – such that people with this gene tend to experience more anger across situations and over time. The L variant is coded on the X chromosome, thus men, more than women, tend to express the L variant. This is because men only have one X chromosome, whereas women have two X chromosomes – meaning if a woman has an L variant on one X chromosome her other X chromosome can counteract the effect.
Further work has found that the presence of this gene combined with specific situations together causes the highest levels of aggression (called a gene X environment interaction). For instance, children with the L variant of MAOA and who had been maltreated showed more antisocial behavior than children with the L variant but no neglect, or children who were neglected but did not have the L variant (Caspi et al., 2002). Other work has found that when participants were excluded in a cyberball game, those participants who took the most points away from another player (a form of aggression) are those with the L variant (Gallardo-Pujol et al., 2013). These findings were not found when participants were included in the game – suggesting the interaction is between the eliciting event (inclusion or exclusion) and biology. Another interesting study (Meyer-Lindenberg et al., 2006) found that when people with the L variant viewed angry and fearful faces, L carriers showed more amygdala activation and less orbitofrontal activation than H variant carriers. This study also looked at how the volume of the amygdala, OFC, cingulate cortex differed for men and women with the L variant. Two important results were found:
- 1) Participants with the L variant (vs. high activity variant) had reductions in the amygdala and cingulate cortex. Reductions in these areas are linked to more aggression.
- 2) Male participants with the L variant showed greater volume of OFC than males with H variant. Females with L and H variant did not show differences in OFC.
Testosterone
Testosterone is a male androgen that is positively correlated with aggression and anger. Because men produce more testosterone than women, this may explain the common gender differences seen in aggression – with men expressing more aggression than women. While playing a cyberball game, male participants’ testosterone increased from baseline to the time when they were excluded from the cyberball game (Peterson & Harmon-Jones, 2012). In fact, men who reported more anger at the end of the game showed higher levels of testosterone! This means testosterone increases proportionately with our subjective feelings of anger.
Although most studies examine testosterone with male participants, one study (van Honk et al., 2001) looked at the effects of testosterone on young female participants’ physiological responses to anger. After taking baseline measures of heart rate, two within-subjects independent variables were manipulated. The first within IV was whether the participants had a testosterone shot or placebo shot. Then, after each shot, all participants viewed three faces with three different emotional expressions – angry, happy, and neutral. So, all participants received the testosterone shot and viewed the three faces, then received the placebo shot and viewed the three faces again, counterbalanced of course. The dependent variable was how much heart rate increased from baseline when viewing each face.
See Figure 3 for a depiction of the findings. When viewing neutral and happy faces, participants did not show an increase in heart rate for the testosterone or placebo shot conditions. When viewing the angry faces, female participants showed an increase in heart after receiving the testosterone shot, but not in the control shot condition. But this difference between the testosterone and placebo shot groups did not occur until three seconds after viewing photo. The authors stated these findings show that testosterone causes SNS activation to angry faces in preparation for aggressive and dominance behaviors. But remember, often when participants view angry faces they might actually experience fear, not anger.
Figure 3
Average heart rate increases for testosterone and placebo conditions after viewing three emotional facial expressions
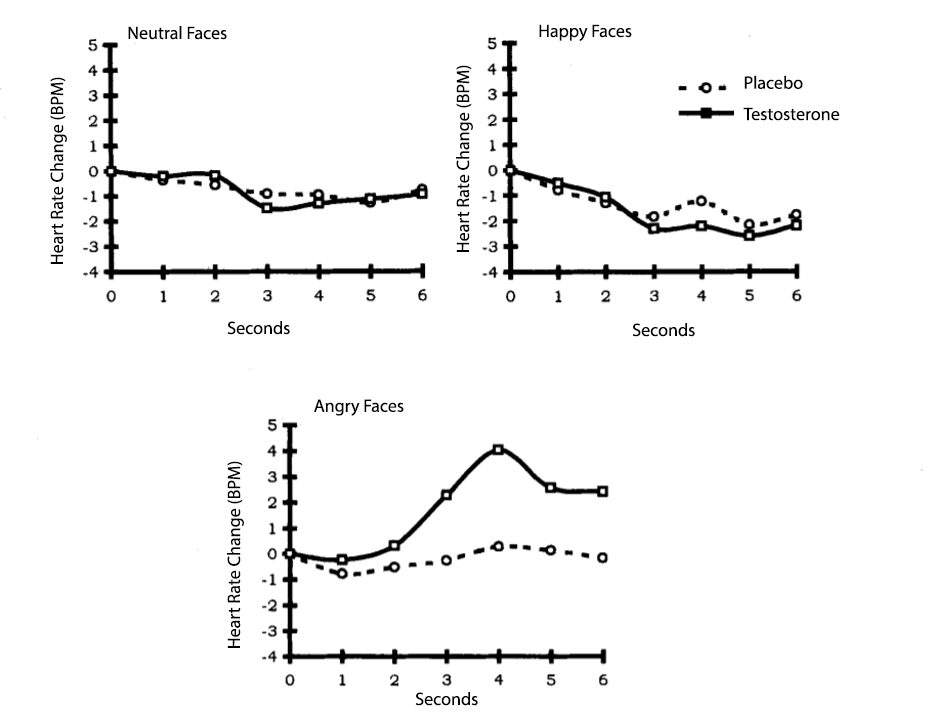
Reproduced from “A single administration of testosterone induces cardiac accelerative responses to angry faces in healthy young women,” by J. van Honk et al., 2001, Behavioral neuroscience, 115(1), p. 240 (A single administration of testosterone induces cardiac accelerative responses to angry faces in healthy young women.). Copyright 2001 by the American Psychological Association.
Matching Activity!
Featured below is a drag and drop activity that lists brain structures on the left side of a table. On the right side of the table are drop zones for emotional changes. Drag and drop the correct emotion changes from the top of the activity into the drop zones for each brain structure.
Note: Some of the emotional changes are used in more than one place, and you can drag more than one element into the drop zones on the right. Additionally, penalties have been turned on for this activity, resulting in a deduction of 1 point for missing or incorrectly dropped text.
Consistency and Discriminable Brain Patterns. In Vytal and Hamann’s (2010) meta-analysis, anger consistently activated the left inferior frontal gyrus (IFG; BA 47) and the right parahippocampal gyrus (PHG; BA 35). (see Figures 4 and 5).
Figure 4
Consistency Patterns for Emotion Anger
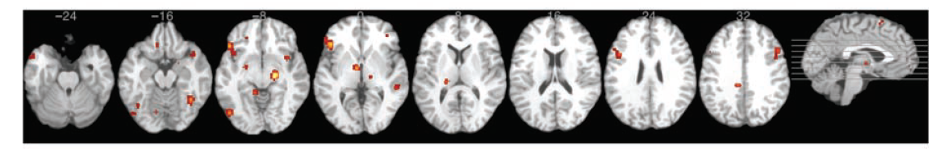
Adapted from “Neuroimaging support for discrete neural correlates of basic emotions: a voxel-based meta-analysis,” by K. Vytal and S. Hamann, 2010, . Journal of Cognitive Neuroscience, 22(12), p. 2870 (Neuroimaging Support for Discrete Neural Correlates of Basic Emotions: A Voxel-based Meta-analysis ). Copyright 2012 by Massachusetts Institute of Technology.
| Comparison | Anger resulted in more activation in… |
|---|---|
| Anger vs. Happiness | IFG (BA 47); Right PHG (BA 35) |
| Anger vs. Sadness | IFG (BA 47); Right PHG (BA 35) |
| Anger vs. Fear | Left IFG (BA 47); right MFG (BA 9) |
| Anger vs. Disgust | Left IFG (BA 47); Left fusiform gyrus (BA 19) |
Note: BA = Brodmann’s Area; IFG = Interior Frontal Gyrus; PHG = Right parahippocampal gyrus; MFG = Middle Frontal Gyrus. Adapted from “Neuroimaging support for discrete neural correlates of basic emotions: a voxel-based meta-analysis,” by K. Vytal and S. Hamann, 2010, . Journal of Cognitive Neuroscience, 22(12), p. 2872 – 2874 (Neuroimaging Support for Discrete Neural Correlates of Basic Emotions: A Voxel-based Meta-analysis ). Copyright 2012 by Massachusetts Institute of Technology.
Figure 5
Brodmann Areas associated with anger
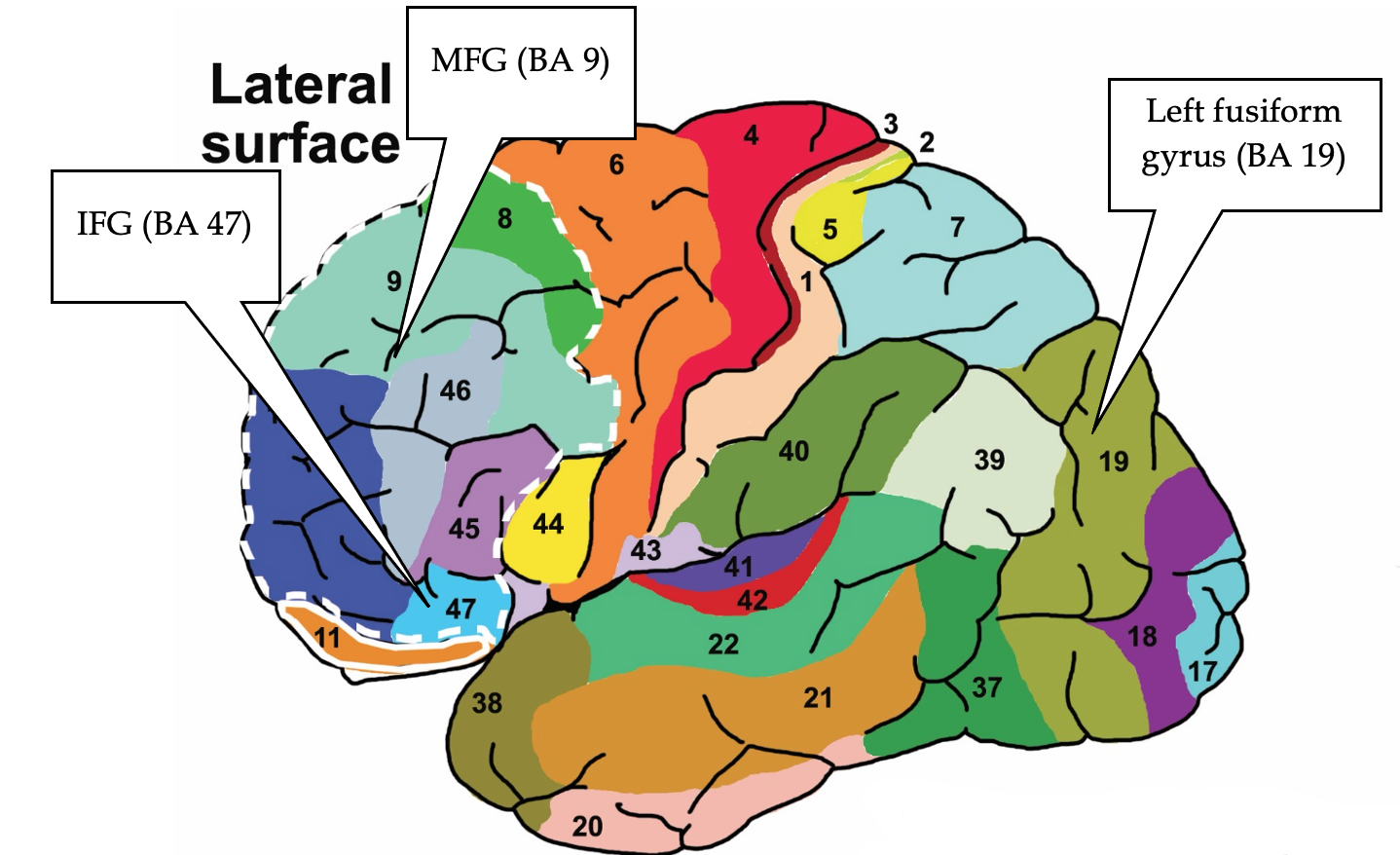
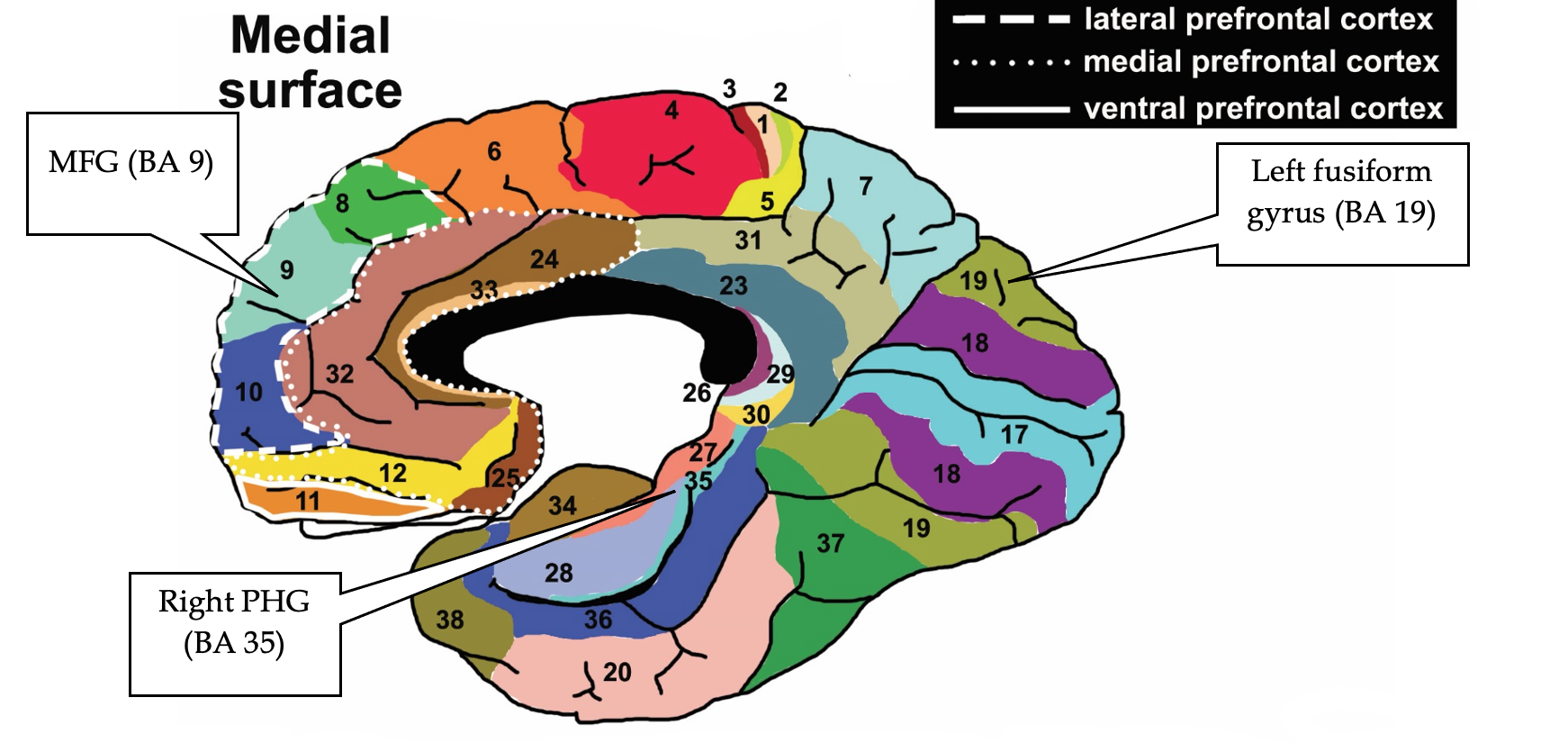
Note. Lateral is an outer view of the brain. Medial is a side view of the brain with the right and left side of the lateral brain divided in half. Reproduced from “Brodmann Areas” by Vysha, 2013. Open Access, Creative Commons CC0 1.0 Universal Public Domain Dedication. Retrieved from File:Brodmann areas.jpg

